
I love this simple activity because there is so much science going on in it, from physics to chemistry, there really is a lot of tidbits you can plop in the conversation while they are playing to help set them up for future success. Plus, dancing corn, or dancing raisins! How cool is that?!?
I also love this project because you don’t need all that much to make it happen. You need popcorn kernels with either baking soda and vinegar or carbonated water.
Bonus if you do both since you can then talk about how the baking soda and vinegar form carbon dioxide, which is what the bubbles in soda water are from!
Fill a large, wide mouth, jar with water and let your kids drop in a few handfuls of popcorn kernels. Then give them cups of baking soda and vinegar that they can mix into the jar to see what happens.
Can they make the corn dance around?
If you are doing the project with popcorn kernels and carbonated water, just fill up the jar with carbonated water and plop the kernels in.

You can also encourage them to try other items to see if they can make them dance. What about cranberries? Raisins? Frozen corn? This activity can be made to fill as much time as you want. Be sure to have some towels on hand for cleanup!
Dr. Erica did a similar experiment in her Pirate’s Treasure Buoyancy lab. You can watch her explain the science here or read about it below!
And just as a note: cranberries and frozen corn float, so they can’t dance! Craisins and corn, however, will dance!
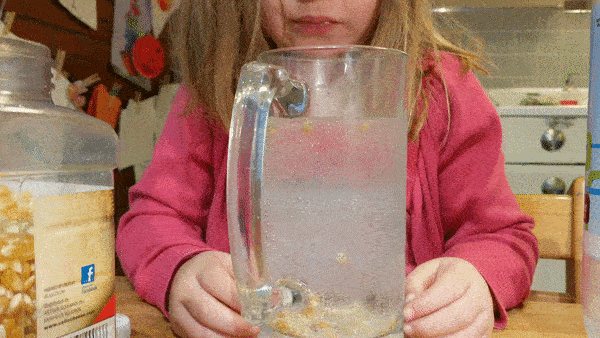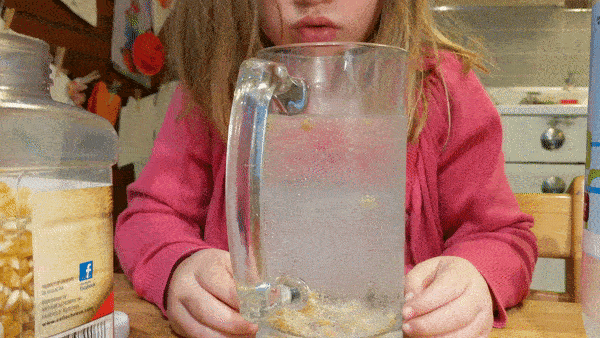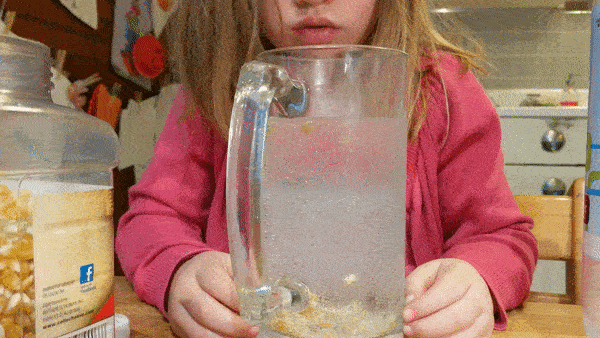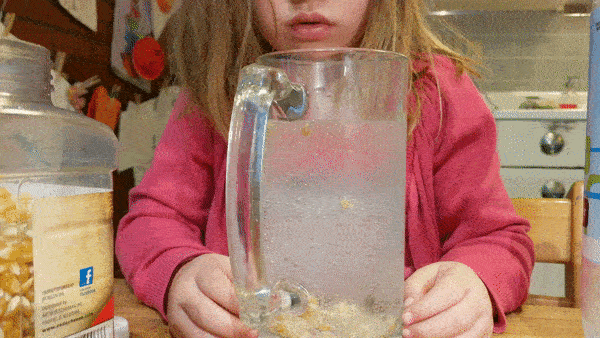
What science is going on behind the scenes? How do dancing corn and dancing raisins work?
Wondering how this could work with either soda water or baking soda and vinegar? It all comes down to physics. Yes, physics…but read on, it’s not that bad!
The physics of dancing corn and dancing raisins – How Newton’s Second law straps on the dancing shoes.
Newton’s second law of motion says that the acceleration of an object is determined by the overall (or net) forces acting on it. In our case, we have three forces acting on the kernels. The force of gravity is pulling it down.
The buoyant force of the kernel is pulling it up, but not very much. And the buoyant forces of the connected carbon dioxide bubbles are pulling the kernel up. So, three forces, one down (gravity) and two up (the buoyant force of the kernel and the carbon dioxide bubbles).
Without the extra buoyant force of the bubbles, gravity would always win and the kernels would always be at the bottom of the cup.
Try this project in water and see for yourself. There would be no dancing corn (or raisins for that matter).
However, the buoyant force of the kernel itself is nearly the force of gravity, they are close, but gravity is always slightly larger and always pulling the corn down (that gravity, what a downer!).
When small bubbles of carbon dioxide attach to the kernel, however, we get lots and lots of tiny little buoyant forces pointing up (lots and lots, for lots and lots of little bubbles). These all add up and grow as more bubbles attach themselves to the corn kernel.
Once the buoyant forces become larger than gravity then the kernels start to float up to the top.
When they hit the top though, some bubbles pop! Oh no! If just enough of those little upward forces pop then gravity will be the winner yet again, and the corn kernel will sink down. And at the bottom, guess what happens? More bubbles attach to the kernel and start to lift it up.
It is an endless cycle of Newton’s second law that creates dancing corn! Well, endless until your bubbles run out! Of course, with a little chemistry, you can keep those bubbles bubbling for a long time.
The chemistry of dancing corn and dancing raisins – how reactions can turn liquids into gas.
Unless you have a Soda Stream at home, you can’t see how carbon dioxide makes its way into water to make it fizzy. If you do, you might know how the process works. A canister of pressurized carbon dioxide gas sits inside the Soda Stream, when you press the button it releases the carbon dioxide.
If you screw a bottle onto the Soda Stream and press the button you are forcing the pressurized carbon dioxide to enter the bottle with the water. The gas dissolves into the water, with more pressure, or colder water, allowing more carbon dioxide to be dissolved.
You can use Henry’s law to discover exactly how much can be dissolved, but I find just blasting the cartridge until I hear the sound of it coming out the top works perfectly well.
Once the carbon dioxide is in the bottle and dissolved into the water pressure needs to stay over it to keep it there, otherwise, it will slowly bubble out. Of course, those bubbles are made of carbon dioxide!
So what about the chemistry of baking soda and vinegar?
Vinegar is a solution of acetic acid that reacts with baking soda (known as sodium bicarbonate), to create sodium acetate and carbonic acid. Carbonic acid is CO3 instead of C02, but is very unstable.
That extra oxygen is very flaky and is always looking for a better group of friends to hang out with. It finds those friends in a hydrogen party. One of the oxygens in carbonic acid goes to hang with two hydrogens to make, drumroll….water!
And of course, if you take an oxygen away you have CO2, also known as carbon dioxide (the di- is a common prefix for two in chemistry!).
Those carbon dioxide bubbles are ready and waiting to attach themselves to anything, including and corn kernels or dried up raisins that might be looking to dance!
The physics of carbon dioxide.
There is another cool thing about carbon dioxide. While it is lighter than water (and thus the bubbles float to the top), it is heavier than air.
That means that it accumulates at the top of the water until it fills up the remaining part of the glass with carbon dioxide gas and overflows the edges.
It would probably look super cool if you could see it right? You can! If you use dry ice that is. Dry ice is solid carbon dioxide, and if you have ever stirred a witches cauldron you have likely seen dry ice gas as it boils away in water. That white vapor always sinks to the floor and cascades over the edges of the cauldron!
Since carbon dioxide is heavier than air it is great for putting out fires, which need air to survive!
Check out our other fun Thanksgiving activities!
- Pumpkin Pie – the science of baking the perfect pie
- Cranberry Ships – engineer ships from cranberries
We just found this and look forward to trying these things to trying these thing, especially the dancing corn. thank you for the sight
Thanks Margaret, and please let me know if you have any questions. My 3-year old played with the dancing corn for hours!