Mesmorize friends with this fun magical chemistry experiment that changes color with the pour of a glass or the breath from your lips!
Jump to a section
Introduction
Project Ingredients
Project Setup
Tips and Tricks
Conclusion
Bring potions class to your home
One of my favorite parts of Harry Potter is Potions class. I love that JK Rowling has gotten a whole generation, and generations to come, insterested in the idea that you can mix different materials together to make something new.
Sure, we won’t be adding leeches and hairs to make a true polyjuice potion in our Harry Potter Potions class, but we will be mixing up a lot of fun! Looking for other potions ideas? Check out our other blog posts!
Rosea Revelantis – Learn about acids, bases, and the power of your breath.
Acids and bases are a fundamental pillar to chemistry. But what are acids and bases?
To learn about acids and bases we first want to think about water – H2O. About 1 in 10 million molecules found in pure water isn’t H2O, however. It is H3O+, a tiny charged molecule. It’s alter ego twin is also floating around in the same concentration, OH-. In fact, H3O+ and OH- can combine to make two normal water molecules – notice how there are 4 H’s and 2 O’s?
Water is considered neutral. There isn’t all that much charge floating around. Sometimes, however, when we mix things, we create a TON of charge in our solution. If the solution has lots of positive charges (like H3O+) then it is acidic, if it has lots of negative charges (like OH-), it is basic.
In this project we will use sodium carbonate to add negative charges to a liquid, our breath to add positive charges to a liquid (what? how can that be?!?), and a pH indicator to show us if the solution is an acid or a base.
Project Ingredients

Kids Science Project Setup
- Sprinkle baking soda on foil and bake at 200 degrees for about 10-15 minutes (find out why you need to bake the baking soda below)
- Sprinkle a small amount of baking soda into a clear jar
- Fill a second clear jar with water and add 2-3 drops of phenolphthalin
- Pour the liquid from the second jar into the first to turn your solution hot pink
- Use straw and bubbles from your breath to turn the solution back to clear (find out how this works below)
- Repeat the process as many times as you like!
How the Harry Potter Potion Rosea Revelantis Works

1. The role of phenolphthalin
Phenolphthalin is called a pH indicator. That means the chemical compound of phenolphthalin is sensitive to how much positive (acidic) and negative (basic) charges are in the solution with it.
When there are more negative charges phenolphthalin is pink. When phenolphthalin is in a solution with more positive charges it is clear. This is because the charges affect what phenolphthalin looks like – it would be like your house changing what it looks like based on the type of weather outside. Here are the two states of phenolphthalin we see in this project – can you spot the differences? These differences are the key to our magical color changes!
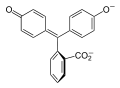
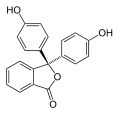
2. The role of baking soda (and why you need to cook it)
Baking soda is also known as sodium bicarbonate. It is slightly basic and much of the reason that our cakes have delightfully airy bubbles to keep in delicious moisture. For this project baking soda is not basic enough to turn our solution pink, we need something more basic.
We need baked soda. Yep, we’re going to make baked soda by baking baking soda. This process squeezes out water and carbon dioxide from the baking soda powder and leaves behind sodium carbonate instead of sodium bicarbonate.
You don’t have to bake it all that long, just 10-15 minutes at 200-300 degress should do it for our purposes, but if you make a bunch you can use it as a flavoring in many chineese recipes!

3. The role of your breath
It might seem odd to have your very own breath used in a chemistry experiment. You don’t have to use your breath, you can use any acid to turn the solution back to acidic, and thus clear. But, seriously? It is so much cooler to make that change through your own actions.
How is your breath acidic? Well, it isn’t really, not until it hits the water. Our breath has a lot of carbon dioxide in it. When carbon dioxide gas mixes with water it creates carboxylic acid. This is actually a really big problem with our oceans and the increase in carbon dioxide in our atmosphere. It is driving carboxylic acid to be created where carbon dioxide in the air meets the water in the ocean. That in turn creates acidified oceans, which our oceanic friends don’t like.
In fact, this project can be a great way to start talking about climate change and our impact on the acidification of the oceans!
Side note: You will have to blow bubbles for a little while to make your solution acidic again. If you added a lot of baked baking soda you will have to blow a lot of bubbles to turn it back to clear.
Once your solution is clear you can add more base (baked baking soda) to make it pink and let the cycle begin again. Just make sure you do this project with older kids that won’t drink the solution, as the phenolphthalin is not edible!
Take it further
Try different things in your house to control the acidity of your solution. What happens if you add something that is very basic, like drain cleaner? Does it turn pink, does it turn a different color? What about if you add something that is very acidic? Does it always stay clear? Or is there another conformation of phenolphthalin at very low pH values (very acidic)?
If you loved this mixing acid/base project you will also love our purple cabbage project!